|
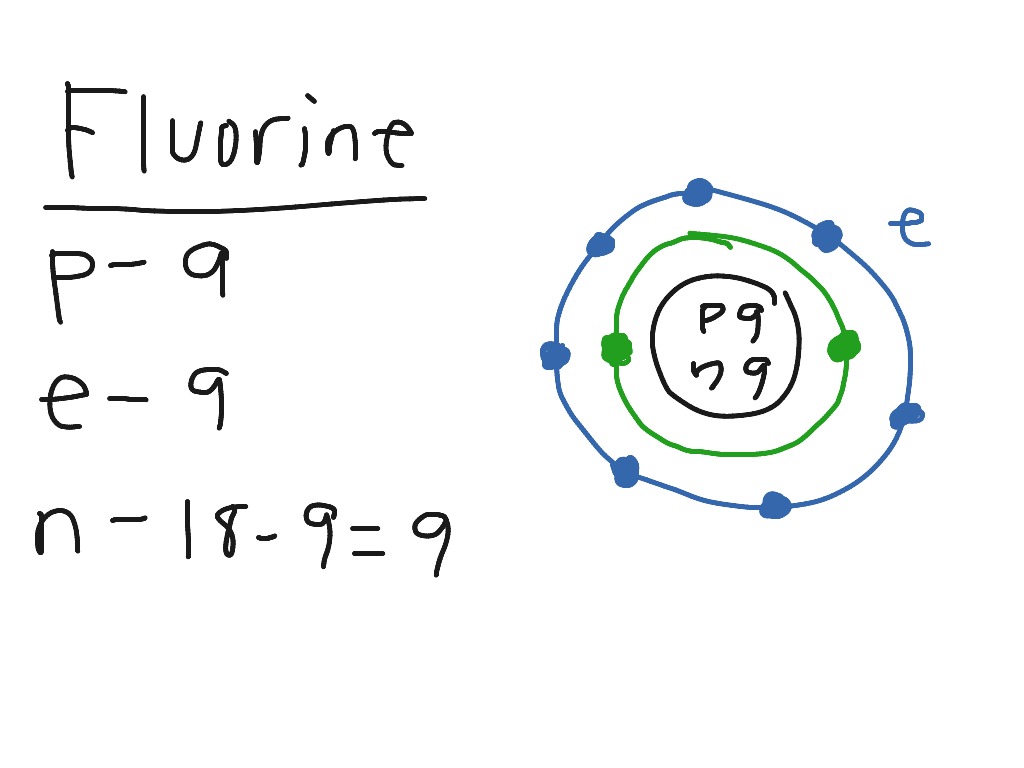
It will violently rip an electron off almost any other atom and is very difficult to isolate.įluorine has 7 valence electrons. The fluorine atom has an electron configuration of 2s 2 2p 5, or 1s 22s 22p 5 and it the most electronegative element on the periodic table, with an electronegativity of 3.98. Fluorine is above chlorine on the periodic table, and is the lightest, most dangerous, and most reactive of all the halogens. It sits at the top of the halogen group, group 17, to the right of oxygen and the left of the element neon. The element fluorine has an atomic symbol of F, and atomic number 9.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
